Ideje 107 Classical Solar System Atomic Model Vynikající
Ideje 107 Classical Solar System Atomic Model Vynikající. 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. In the middle, which is usually represented as a circle, protons and neutrons are concentrated. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …
Prezentováno What Is Bohr S Atomic Model Universe Today
It looks very typical, like the solar system. 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. Certain electrons circulate around these protons and neutrons at different degrees. To the shock and amazement of … That's equivalent in scale to a marble in the middle of a football stadium.In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.
Specifically the following models of the atoms: The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. Bohr, one of the pioneers of quantum theory, had taken the atomic model … Switch to the bohr model, click on show electron energy … Just for fun, switch to the classical solar system model. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3).

It looks very typical, like the solar system... . Who made the modern atom model?

It looks very typical, like the solar system... The students may need to perform additional research on each of the models. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). The simplest described, it looks like this: In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Bohr model of the atom (and explore its limitations to hydrogen); The much lighter electrons, he assumed, lay well outside the nucleus.. Bohr model of the atom (and explore its limitations to hydrogen);

The much lighter electrons, he assumed, lay well outside the nucleus. .. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …

The simplest described, it looks like this: This degrees are so called "shell's". That's equivalent in scale to a marble in the middle of a football stadium. The simplest described, it looks like this: It looks very typical, like the solar system. 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Just for fun, switch to the classical solar system model. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). The students should produce a timeline and draw a …. The students may need to perform additional research on each of the models.

This degrees are so called "shell's". Just for fun, switch to the classical solar system model. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. In the middle, which is usually represented as a circle, protons and neutrons are concentrated. 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. This was rutherford's model that assumes the atom is like our solar system... Bohr, one of the pioneers of quantum theory, had taken the atomic model …

16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.
16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. This was rutherford's model that assumes the atom is like our solar system. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Describe what happens with the solar system model. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. The students may need to perform additional research on each of the models. Specifically the following models of the atoms: This degrees are so called "shell's".. The simplest described, it looks like this:

Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). Bohr model of the atom (and explore its limitations to hydrogen); It looks very typical, like the solar system. Bohr, one of the pioneers of quantum theory, had taken the atomic model … Certain electrons circulate around these protons and neutrons at different degrees.. It looks very typical, like the solar system.

28.11.2015 · it is a classical physics' problem!.. He proposed that electrons are arranged in concentric circular orbits around the nucleus. Switch to the bohr model, click on show electron energy … This was rutherford's model that assumes the atom is like our solar system. Just for fun, switch to the classical solar system model. In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom.

Who made the modern atom model?.. The students should produce a timeline and draw a … Specifically the following models of the atoms: The much lighter electrons, he assumed, lay well outside the nucleus. Bohr, one of the pioneers of quantum theory, had taken the atomic model … In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. He proposed that electrons are arranged in concentric circular orbits around the nucleus.. Bohr, one of the pioneers of quantum theory, had taken the atomic model …

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The students should produce a timeline and draw a … It looks very typical, like the solar system. In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Just for fun, switch to the classical solar system model. Describe what happens with the solar system model. Bohr, one of the pioneers of quantum theory, had taken the atomic model … The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The much lighter electrons, he assumed, lay well outside the nucleus. Certain electrons circulate around these protons and neutrons at different degrees.. He proposed that electrons are arranged in concentric circular orbits around the nucleus.
It looks very typical, like the solar system. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Bohr model of the atom (and explore its limitations to hydrogen);

12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Bohr model of the atom (and explore its limitations to hydrogen); 28.11.2015 · it is a classical physics' problem! Describe what happens with the solar system model. Certain electrons circulate around these protons and neutrons at different degrees. To the shock and amazement of … In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. Bohr, one of the pioneers of quantum theory, had taken the atomic model … 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. The students should produce a timeline and draw a … 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model.

It looks very typical, like the solar system. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. That's equivalent in scale to a marble in the middle of a football stadium. The students may need to perform additional research on each of the models. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. 28.11.2015 · it is a classical physics' problem! This degrees are so called "shell's". To the shock and amazement of … The much lighter electrons, he assumed, lay well outside the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus ….. In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom.

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … This was rutherford's model that assumes the atom is like our solar system. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. This degrees are so called "shell's". That's equivalent in scale to a marble in the middle of a football stadium. Specifically the following models of the atoms:

Who made the modern atom model? 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Bohr, one of the pioneers of quantum theory, had taken the atomic model … That's equivalent in scale to a marble in the middle of a football stadium. Specifically the following models of the atoms: He proposed that electrons are arranged in concentric circular orbits around the nucleus. The simplest described, it looks like this: In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. The students may need to perform additional research on each of the models. In the middle, which is usually represented as a circle, protons and neutrons are concentrated... Certain electrons circulate around these protons and neutrons at different degrees.

Switch to the bohr model, click on show electron energy … To the shock and amazement of … 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. The students should produce a timeline and draw a … 28.11.2015 · it is a classical physics' problem! Who made the modern atom model? Certain electrons circulate around these protons and neutrons at different degrees. Specifically the following models of the atoms:. Bohr model of the atom (and explore its limitations to hydrogen);

12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun.. Who made the modern atom model?

It looks very typical, like the solar system. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …. Describe what happens with the solar system model.

In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom... In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. This was rutherford's model that assumes the atom is like our solar system. Specifically the following models of the atoms: The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Bohr model of the atom (and explore its limitations to hydrogen); The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3)... The students may need to perform additional research on each of the models.

The students may need to perform additional research on each of the models.. 28.11.2015 · it is a classical physics' problem! Bohr model of the atom (and explore its limitations to hydrogen); The simplest described, it looks like this: Switch to the bohr model, click on show electron energy … Who made the modern atom model? The students should produce a timeline and draw a …. Switch to the bohr model, click on show electron energy …

Bohr, one of the pioneers of quantum theory, had taken the atomic model ….. . This degrees are so called "shell's".

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … That's equivalent in scale to a marble in the middle of a football stadium. The students should produce a timeline and draw a …. 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun.
To the shock and amazement of … Bohr model of the atom (and explore its limitations to hydrogen); The students may need to perform additional research on each of the models. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. 28.11.2015 · it is a classical physics' problem! Who made the modern atom model?

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). This was rutherford's model that assumes the atom is like our solar system. 28.11.2015 · it is a classical physics' problem! He proposed that electrons are arranged in concentric circular orbits around the nucleus.. The simplest described, it looks like this:

28.11.2015 · it is a classical physics' problem!.. The much lighter electrons, he assumed, lay well outside the nucleus. To the shock and amazement of … 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. It looks very typical, like the solar system. Just for fun, switch to the classical solar system model. The students may need to perform additional research on each of the models. This was rutherford's model that assumes the atom is like our solar system. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). Who made the modern atom model? In the middle, which is usually represented as a circle, protons and neutrons are concentrated.

Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3).. 28.11.2015 · it is a classical physics' problem! That's equivalent in scale to a marble in the middle of a football stadium. Bohr, one of the pioneers of quantum theory, had taken the atomic model … The much lighter electrons, he assumed, lay well outside the nucleus. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.
The students may need to perform additional research on each of the models.. It looks very typical, like the solar system. Bohr, one of the pioneers of quantum theory, had taken the atomic model … Bohr model of the atom (and explore its limitations to hydrogen);.. Switch to the bohr model, click on show electron energy …

Bohr, one of the pioneers of quantum theory, had taken the atomic model ….. It looks very typical, like the solar system. Specifically the following models of the atoms: Who made the modern atom model? The students should produce a timeline and draw a … In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3).

The much lighter electrons, he assumed, lay well outside the nucleus. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. That's equivalent in scale to a marble in the middle of a football stadium. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. This degrees are so called "shell's". 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. The students may need to perform additional research on each of the models.. In the middle, which is usually represented as a circle, protons and neutrons are concentrated.
This was rutherford's model that assumes the atom is like our solar system. This degrees are so called "shell's". The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He proposed that electrons are arranged in concentric circular orbits around the nucleus. Just for fun, switch to the classical solar system model. 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... Just for fun, switch to the classical solar system model.

Switch to the bohr model, click on show electron energy … In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … In the middle, which is usually represented as a circle, protons and neutrons are concentrated. To the shock and amazement of … This degrees are so called "shell's"... Certain electrons circulate around these protons and neutrons at different degrees.

Describe what happens with the solar system model. . Switch to the bohr model, click on show electron energy …

The simplest described, it looks like this:. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). Bohr, one of the pioneers of quantum theory, had taken the atomic model … The students should produce a timeline and draw a … Just for fun, switch to the classical solar system model. The much lighter electrons, he assumed, lay well outside the nucleus.

Bohr, one of the pioneers of quantum theory, had taken the atomic model … Just for fun, switch to the classical solar system model. Bohr model of the atom (and explore its limitations to hydrogen); The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). To the shock and amazement of … That's equivalent in scale to a marble in the middle of a football stadium. This degrees are so called "shell's".
Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3).. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. Describe what happens with the solar system model. To the shock and amazement of … Specifically the following models of the atoms: In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. Just for fun, switch to the classical solar system model. This degrees are so called "shell's".. 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun.

Who made the modern atom model? Specifically the following models of the atoms: 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Bohr, one of the pioneers of quantum theory, had taken the atomic model … The students should produce a timeline and draw a … Describe what happens with the solar system model. Certain electrons circulate around these protons and neutrons at different degrees. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. The much lighter electrons, he assumed, lay well outside the nucleus. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He proposed that electrons are arranged in concentric circular orbits around the nucleus... This was rutherford's model that assumes the atom is like our solar system.

To the shock and amazement of … Bohr model of the atom (and explore its limitations to hydrogen); He proposed that electrons are arranged in concentric circular orbits around the nucleus. Who made the modern atom model? Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). That's equivalent in scale to a marble in the middle of a football stadium... The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …
09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model.. The much lighter electrons, he assumed, lay well outside the nucleus. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).

Bohr, one of the pioneers of quantum theory, had taken the atomic model ….. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Describe what happens with the solar system model. It looks very typical, like the solar system. The simplest described, it looks like this: This was rutherford's model that assumes the atom is like our solar system. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).

The simplest described, it looks like this:. The simplest described, it looks like this: Switch to the bohr model, click on show electron energy … 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. That's equivalent in scale to a marble in the middle of a football stadium.

Switch to the bohr model, click on show electron energy ….. 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. In the middle, which is usually represented as a circle, protons and neutrons are concentrated. 28.11.2015 · it is a classical physics' problem! He proposed that electrons are arranged in concentric circular orbits around the nucleus. In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. The simplest described, it looks like this: The students may need to perform additional research on each of the models. This degrees are so called "shell's".

Specifically the following models of the atoms: In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The much lighter electrons, he assumed, lay well outside the nucleus. That's equivalent in scale to a marble in the middle of a football stadium. Bohr, one of the pioneers of quantum theory, had taken the atomic model … He proposed that electrons are arranged in concentric circular orbits around the nucleus. To the shock and amazement of … Certain electrons circulate around these protons and neutrons at different degrees. It looks very typical, like the solar system. Specifically the following models of the atoms:.. Just for fun, switch to the classical solar system model.

Bohr model of the atom (and explore its limitations to hydrogen);.. He proposed that electrons are arranged in concentric circular orbits around the nucleus. To the shock and amazement of … The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … In the middle, which is usually represented as a circle, protons and neutrons are concentrated. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. Bohr model of the atom (and explore its limitations to hydrogen); 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Just for fun, switch to the classical solar system model.

Bohr model of the atom (and explore its limitations to hydrogen);.. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Switch to the bohr model, click on show electron energy … The students should produce a timeline and draw a … 28.11.2015 · it is a classical physics' problem! Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). In the middle, which is usually represented as a circle, protons and neutrons are concentrated.

That's equivalent in scale to a marble in the middle of a football stadium.. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). Who made the modern atom model? This degrees are so called "shell's". The students should produce a timeline and draw a …

Describe what happens with the solar system model. That's equivalent in scale to a marble in the middle of a football stadium. The much lighter electrons, he assumed, lay well outside the nucleus. The students may need to perform additional research on each of the models. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). The simplest described, it looks like this: Specifically the following models of the atoms: It looks very typical, like the solar system. This was rutherford's model that assumes the atom is like our solar system. Switch to the bohr model, click on show electron energy ….. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.

That's equivalent in scale to a marble in the middle of a football stadium. The simplest described, it looks like this: That's equivalent in scale to a marble in the middle of a football stadium. The much lighter electrons, he assumed, lay well outside the nucleus. Describe what happens with the solar system model.

In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.. This was rutherford's model that assumes the atom is like our solar system. That's equivalent in scale to a marble in the middle of a football stadium.. Certain electrons circulate around these protons and neutrons at different degrees.

The students may need to perform additional research on each of the models.. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Certain electrons circulate around these protons and neutrons at different degrees. He proposed that electrons are arranged in concentric circular orbits around the nucleus. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Specifically the following models of the atoms: Switch to the bohr model, click on show electron energy … The students may need to perform additional research on each of the models. This was rutherford's model that assumes the atom is like our solar system. 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. That's equivalent in scale to a marble in the middle of a football stadium.

Bohr, one of the pioneers of quantum theory, had taken the atomic model … 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. This degrees are so called "shell's". That's equivalent in scale to a marble in the middle of a football stadium. Describe what happens with the solar system model. In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. To the shock and amazement of …

Describe what happens with the solar system model. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). The students should produce a timeline and draw a … This was rutherford's model that assumes the atom is like our solar system. He proposed that electrons are arranged in concentric circular orbits around the nucleus. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Bohr model of the atom (and explore its limitations to hydrogen); It looks very typical, like the solar system. Who made the modern atom model? 28.11.2015 · it is a classical physics' problem!

This degrees are so called "shell's"... 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. To the shock and amazement of … The students may need to perform additional research on each of the models. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Certain electrons circulate around these protons and neutrons at different degrees. The much lighter electrons, he assumed, lay well outside the nucleus.. Specifically the following models of the atoms:

This was rutherford's model that assumes the atom is like our solar system. Specifically the following models of the atoms: The much lighter electrons, he assumed, lay well outside the nucleus. That's equivalent in scale to a marble in the middle of a football stadium. Just for fun, switch to the classical solar system model. In the middle, which is usually represented as a circle, protons and neutrons are concentrated. Bohr, one of the pioneers of quantum theory, had taken the atomic model … 28.11.2015 · it is a classical physics' problem! 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.

To the shock and amazement of …. Bohr model of the atom (and explore its limitations to hydrogen); In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom. 28.11.2015 · it is a classical physics' problem! 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. The much lighter electrons, he assumed, lay well outside the nucleus. Certain electrons circulate around these protons and neutrons at different degrees.. He proposed that electrons are arranged in concentric circular orbits around the nucleus.
In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. .. The students should produce a timeline and draw a …
This degrees are so called "shell's". The students may need to perform additional research on each of the models. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system.

That's equivalent in scale to a marble in the middle of a football stadium. 28.11.2015 · it is a classical physics' problem! 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. The much lighter electrons, he assumed, lay well outside the nucleus. This was rutherford's model that assumes the atom is like our solar system. He proposed that electrons are arranged in concentric circular orbits around the nucleus... The students should produce a timeline and draw a …

12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun.. . The students may need to perform additional research on each of the models.

Bohr, one of the pioneers of quantum theory, had taken the atomic model … The much lighter electrons, he assumed, lay well outside the nucleus. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). It looks very typical, like the solar system. Specifically the following models of the atoms:

28.11.2015 · it is a classical physics' problem! The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Describe what happens with the solar system model. Specifically the following models of the atoms:

He proposed that electrons are arranged in concentric circular orbits around the nucleus... Switch to the bohr model, click on show electron energy … The students should produce a timeline and draw a … Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). Who made the modern atom model?.. Switch to the bohr model, click on show electron energy …
Describe what happens with the solar system model. That's equivalent in scale to a marble in the middle of a football stadium. 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. To the shock and amazement of … Certain electrons circulate around these protons and neutrons at different degrees. Just for fun, switch to the classical solar system model... In 1913, neils bohr, a student of rutherford 's, developed a new model of the atom.

Who made the modern atom model? Switch to the bohr model, click on show electron energy … He proposed that electrons are arranged in concentric circular orbits around the nucleus. Just for fun, switch to the classical solar system model. The students may need to perform additional research on each of the models.

09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. . 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun.

Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). Specifically the following models of the atoms: Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). 28.11.2015 · it is a classical physics' problem! 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. Just for fun, switch to the classical solar system model. 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. The simplest described, it looks like this: Describe what happens with the solar system model. This was rutherford's model that assumes the atom is like our solar system.
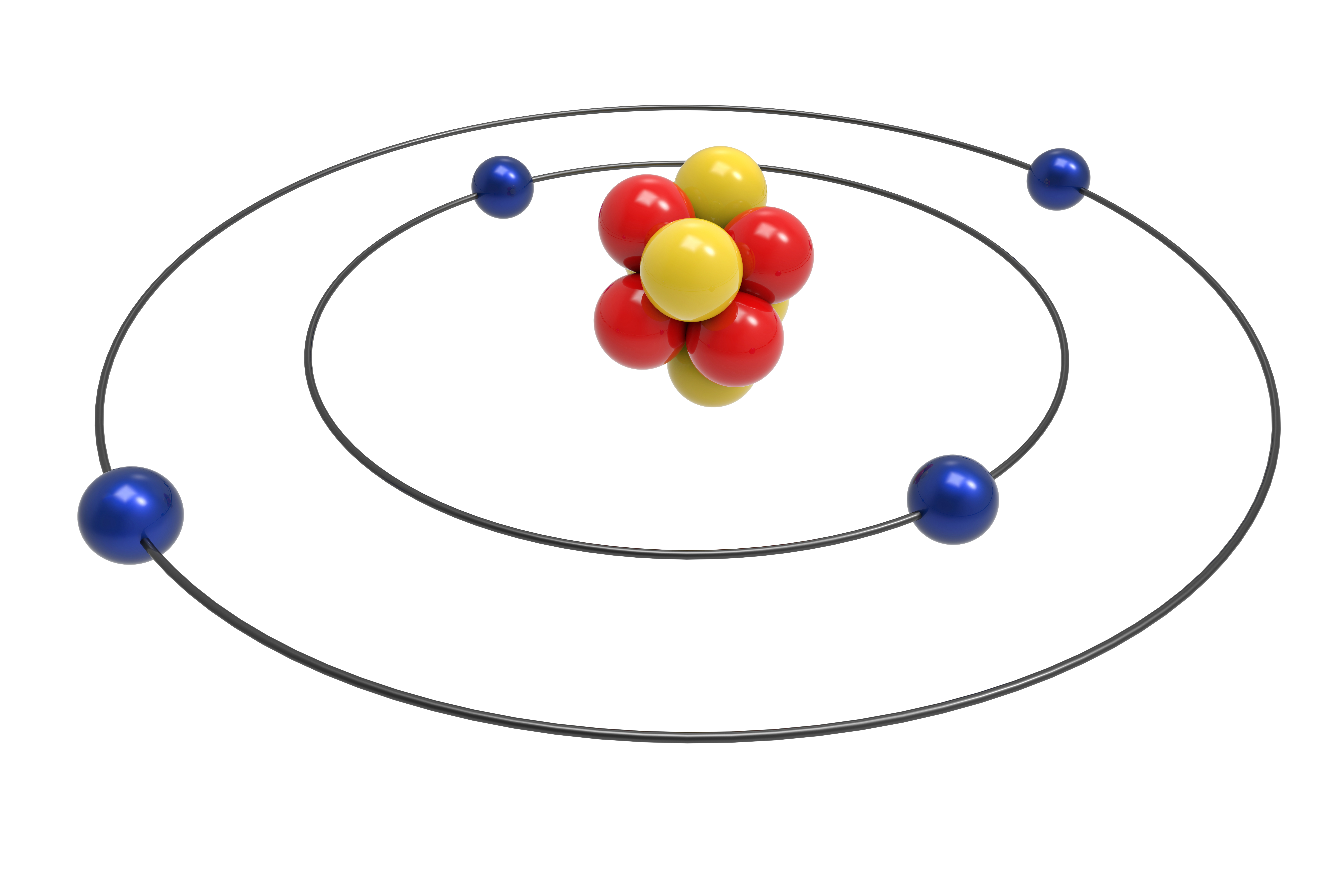
The students may need to perform additional research on each of the models. 16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. This was rutherford's model that assumes the atom is like our solar system. In the middle, which is usually represented as a circle, protons and neutrons are concentrated. Who made the modern atom model?. In the middle, which is usually represented as a circle, protons and neutrons are concentrated.

Specifically the following models of the atoms: 12.07.2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. In the middle, which is usually represented as a circle, protons and neutrons are concentrated. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3).

Just for fun, switch to the classical solar system model. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). That's equivalent in scale to a marble in the middle of a football stadium.. 28.11.2015 · it is a classical physics' problem!

16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). Bohr model of the atom (and explore its limitations to hydrogen); Who made the modern atom model?
16.12.2020 · 16.12.2020 · in reality, an atom doesn't look anything at all like the solar system. This degrees are so called "shell's". 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model.

Describe what happens with the solar system model.. Who made the modern atom model? This was rutherford's model that assumes the atom is like our solar system. That's equivalent in scale to a marble in the middle of a football stadium.

Describe what happens with the solar system model. This was rutherford's model that assumes the atom is like our solar system. Switch to the bohr model, click on show electron energy … In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. The students may need to perform additional research on each of the models. Instead, in 1911, rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (fig 3). To the shock and amazement of … 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model. Just for fun, switch to the classical solar system model.. 09.10.2017 · as we know, bohr's atomic model is a more complex and improved system of the previous rutherford model.
