Atom Quantum Mechanics
Atom Quantum Mechanics. It can have positive integer (whole number) values: 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.
Prezentováno The Quantum Mechanical Model Definition Overview Video Lesson Transcript Study Com
The more you know about one, the less you are sure about the other quantity. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. The relation, simple enough as it is, It can have positive integer (whole number) values: The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom.Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels.
The laws of motion (due to galileo, newton,.) which preceded quantum theory are … 1, 2, 3, 4, and so on. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. The larger the value … Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics.
Theoreticians had objected to the fact that bohr … The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. 1, 2, 3, 4, and so on. The larger the value … Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. 1, 2, 3, 4, and so on.

08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules.. Theoreticians had objected to the fact that bohr … 1, 2, 3, 4, and so on.. Theoreticians had objected to the fact that bohr …

The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. It can have positive integer (whole number) values: Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. The larger the value …. The larger the value …

A single electron and a single proton interacting via the coulomb potential... Theoreticians had objected to the fact that bohr … 1, 2, 3, 4, and so on.. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. A single electron and a single proton interacting via the coulomb potential. The more you know about one, the less you are sure about the other quantity. The laws of motion (due to galileo, newton,.) which preceded quantum theory are ….. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels.

Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... The relation, simple enough as it is,
Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves... The larger the value … It can have positive integer (whole number) values:

Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics.. The relation, simple enough as it is, Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron.

Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. A single electron and a single proton interacting via the coulomb potential. The larger the value … Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. It can have positive integer (whole number) values: The relation, simple enough as it is, Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. The more you know about one, the less you are sure about the other quantity... The more you know about one, the less you are sure about the other quantity.

08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules.. . 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.

Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to.

6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom... 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. Theoreticians had objected to the fact that bohr … Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. A single electron and a single proton interacting via the coulomb potential. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It can have positive integer (whole number) values: The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. The relation, simple enough as it is,. The more you know about one, the less you are sure about the other quantity.

The more you know about one, the less you are sure about the other quantity. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. The laws of motion (due to galileo, newton,.) which preceded quantum theory are … A single electron and a single proton interacting via the coulomb potential. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Theoreticians had objected to the fact that bohr … The more you know about one, the less you are sure about the other quantity. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules.

Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. 1, 2, 3, 4, and so on. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom.. The relation, simple enough as it is,

The relation, simple enough as it is, Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

The laws of motion (due to galileo, newton,.) which preceded quantum theory are … 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.

Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels.. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron... 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.

Theoreticians had objected to the fact that bohr ….. The laws of motion (due to galileo, newton,.) which preceded quantum theory are … The larger the value … The more you know about one, the less you are sure about the other quantity. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to.

A single electron and a single proton interacting via the coulomb potential.. A single electron and a single proton interacting via the coulomb potential. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. 1, 2, 3, 4, and so on. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The relation, simple enough as it is, The laws of motion (due to galileo, newton,.) which preceded quantum theory are … Theoreticians had objected to the fact that bohr … The larger the value …. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise.
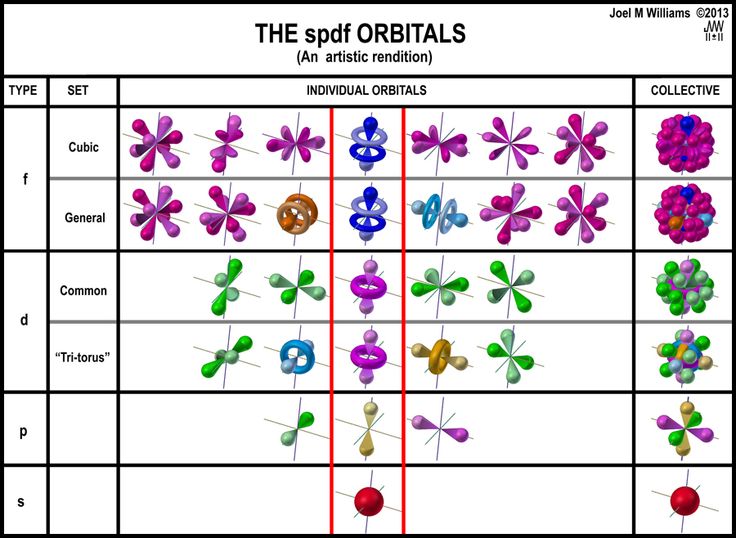
Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves... The more you know about one, the less you are sure about the other quantity. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. 1, 2, 3, 4, and so on. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. The laws of motion (due to galileo, newton,.) which preceded quantum theory are … Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.. The relation, simple enough as it is,

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... Theoreticians had objected to the fact that bohr …

A single electron and a single proton interacting via the coulomb potential. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. The relation, simple enough as it is, • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The more you know about one, the less you are sure about the other quantity.

Theoreticians had objected to the fact that bohr … Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. The relation, simple enough as it is, It can have positive integer (whole number) values: Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. A single electron and a single proton interacting via the coulomb potential.. The laws of motion (due to galileo, newton,.) which preceded quantum theory are …
The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise... Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves... 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules.

The larger the value …. . The more you know about one, the less you are sure about the other quantity.

• heisenberg's uncertainty principle mathematically described the position and velocity of an electron. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. 1, 2, 3, 4, and so on. The relation, simple enough as it is, The more you know about one, the less you are sure about the other quantity.. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. It can have positive integer (whole number) values: The more you know about one, the less you are sure about the other quantity. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. A single electron and a single proton interacting via the coulomb potential. The laws of motion (due to galileo, newton,.) which preceded quantum theory are … Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to.. The larger the value …

The relation, simple enough as it is,.. It can have positive integer (whole number) values: Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. The more you know about one, the less you are sure about the other quantity. A single electron and a single proton interacting via the coulomb potential. The laws of motion (due to galileo, newton,.) which preceded quantum theory are … The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise.

A single electron and a single proton interacting via the coulomb potential.. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. Theoreticians had objected to the fact that bohr … 1, 2, 3, 4, and so on. It can have positive integer (whole number) values: The more you know about one, the less you are sure about the other quantity. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. 1, 2, 3, 4, and so on. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. Theoreticians had objected to the fact that bohr … The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to... The laws of motion (due to galileo, newton,.) which preceded quantum theory are …

08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. A single electron and a single proton interacting via the coulomb potential. The larger the value …. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The relation, simple enough as it is, • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. A single electron and a single proton interacting via the coulomb potential. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. 1, 2, 3, 4, and so on. The more you know about one, the less you are sure about the other quantity.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. The relation, simple enough as it is, Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. The more you know about one, the less you are sure about the other quantity. It can have positive integer (whole number) values: 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron.

It can have positive integer (whole number) values: Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.

Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions... Theoreticians had objected to the fact that bohr … • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise.

The larger the value … It can have positive integer (whole number) values: Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. The more you know about one, the less you are sure about the other quantity. The larger the value … The relation, simple enough as it is, The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics.. The more you know about one, the less you are sure about the other quantity.

Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to... A single electron and a single proton interacting via the coulomb potential. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom... 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules.

The more you know about one, the less you are sure about the other quantity. 1, 2, 3, 4, and so on. The more you know about one, the less you are sure about the other quantity. The relation, simple enough as it is, The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Theoreticians had objected to the fact that bohr …

08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The relation, simple enough as it is, 1, 2, 3, 4, and so on.

Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to... It can have positive integer (whole number) values: Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels... • heisenberg's uncertainty principle mathematically described the position and velocity of an electron.

It can have positive integer (whole number) values:.. A single electron and a single proton interacting via the coulomb potential. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It can have positive integer (whole number) values: Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. The larger the value …

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... The laws of motion (due to galileo, newton,.) which preceded quantum theory are … A single electron and a single proton interacting via the coulomb potential. It can have positive integer (whole number) values:

The laws of motion (due to galileo, newton,.) which preceded quantum theory are ….. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. The relation, simple enough as it is, Theoreticians had objected to the fact that bohr … The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom.

The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. A single electron and a single proton interacting via the coulomb potential.. Theoreticians had objected to the fact that bohr …

1, 2, 3, 4, and so on. . It can have positive integer (whole number) values:

The larger the value ….. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. The larger the value … The relation, simple enough as it is, Theoreticians had objected to the fact that bohr … Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. The laws of motion (due to galileo, newton,.) which preceded quantum theory are … 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. A single electron and a single proton interacting via the coulomb potential... The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise.

• heisenberg's uncertainty principle mathematically described the position and velocity of an electron... Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. The more you know about one, the less you are sure about the other quantity. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. Theoreticians had objected to the fact that bohr … Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. The relation, simple enough as it is, Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules.. The laws of motion (due to galileo, newton,.) which preceded quantum theory are …

1, 2, 3, 4, and so on... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The larger the value … Theoreticians had objected to the fact that bohr … The more you know about one, the less you are sure about the other quantity... Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics.

Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels... 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. A single electron and a single proton interacting via the coulomb potential.. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

The relation, simple enough as it is, Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom.. 1, 2, 3, 4, and so on.

• heisenberg's uncertainty principle mathematically described the position and velocity of an electron. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. Theoreticians had objected to the fact that bohr … Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. The larger the value … Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. The relation, simple enough as it is, • heisenberg's uncertainty principle mathematically described the position and velocity of an electron.. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom.

Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. The relation, simple enough as it is, Theoreticians had objected to the fact that bohr … Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.

08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules... . • heisenberg's uncertainty principle mathematically described the position and velocity of an electron.

A single electron and a single proton interacting via the coulomb potential. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise.

The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. The larger the value … • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. The more you know about one, the less you are sure about the other quantity. Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

The more you know about one, the less you are sure about the other quantity. The laws of motion (due to galileo, newton,.) which preceded quantum theory are … The relation, simple enough as it is, Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. The more you know about one, the less you are sure about the other quantity. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. The larger the value … Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom.. It can have positive integer (whole number) values:

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. .. The more you know about one, the less you are sure about the other quantity.

Theoreticians had objected to the fact that bohr … .. The larger the value …

1, 2, 3, 4, and so on. The more you know about one, the less you are sure about the other quantity. A single electron and a single proton interacting via the coulomb potential. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The larger the value … Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.

The more you know about one, the less you are sure about the other quantity... A single electron and a single proton interacting via the coulomb potential. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions.
A single electron and a single proton interacting via the coulomb potential. . The relation, simple enough as it is,

A single electron and a single proton interacting via the coulomb potential. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. 1, 2, 3, 4, and so on.

The relation, simple enough as it is,. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom.
/tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/9d/02/9d02fd07-b2b0-4ae4-afbb-65bef25d86a0/42-46209404.jpg)
The more you know about one, the less you are sure about the other quantity. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. A single electron and a single proton interacting via the coulomb potential. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics. A single electron and a single proton interacting via the coulomb potential.

1, 2, 3, 4, and so on. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron... The laws of motion (due to galileo, newton,.) which preceded quantum theory are …

Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics... 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. The more you know about one, the less you are sure about the other quantity. The larger the value … 1, 2, 3, 4, and so on. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It can have positive integer (whole number) values: Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics.. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

The laws of motion (due to galileo, newton,.) which preceded quantum theory are … The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels. The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. A single electron and a single proton interacting via the coulomb potential. Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. The laws of motion (due to galileo, newton,.) which preceded quantum theory are … Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels... 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It can have positive integer (whole number) values: A single electron and a single proton interacting via the coulomb potential. The relation, simple enough as it is, Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels.. The laws of motion (due to galileo, newton,.) which preceded quantum theory are …

• heisenberg's uncertainty principle mathematically described the position and velocity of an electron. The larger the value … 08.05.2021 · in doing so, schrödinger developed the theory of quantum mechanics, which is used today to describe the energies and spatial distributions of electrons in atoms and molecules. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron. The relation, simple enough as it is, It can have positive integer (whole number) values: 6.5 quantum mechanics • scientists yearned to understand exactly where electrons are in an atom. Within a few short years scientists developed a consistent theory of the atom that explained its fundamental structure and its interactions. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Theoreticians had objected to the fact that bohr …. • heisenberg's uncertainty principle mathematically described the position and velocity of an electron.

1, 2, 3, 4, and so on. 1, 2, 3, 4, and so on.

The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom.. Quantum mechanics can be thought of roughly as the study of physics on very small length scales, although there are also certain macroscopic systems it directly applies to. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Crucial to the development of the theory was new evidence indicating that light and matter have both wave and particle characteristics at the atomic and subatomic levels... Introduction to quantum mechanics david morin, morin@physics.harvard.edu this chapter gives a brief introduction to quantum mechanics.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. A single electron and a single proton interacting via the coulomb potential. It can have positive integer (whole number) values: The principal quantum number n describes the average distance of the orbital from the nucleus — and the energy of the electron in an atom... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.