104 Quantum Mechanical Model Of Hydrogen Atom
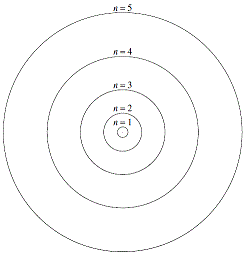
104 Quantum Mechanical Model Of Hydrogen Atom. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.
Tady The Quantum Mechanical Model Of The Atom Ppt Download
Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry An electron in classical and quantum physics do not have the. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron.Quantum mechanics and the atom.
Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. The loss of a photon is shown for the electronic transition with an energy of hf. The energy increases and thus levels are closer to each other. Kaypeeoh72z and 6 more users found this answer helpful. As the n level increases. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics.
Paradoxes of the rutherford model... Equations are derived which correspond to the relativistic mass and the quantum. Orbitals for the hydrogen atom. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry An electron in classical and quantum physics do not have the. Kaypeeoh72z and 6 more users found this answer helpful. Quantum mechanics and the atom. Cited from the book "the stability of matter: How many orbitals are possible at the 3rd energy level (n=3)? The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise.. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics.

Draw the shape of an s and a p orbital. Kaypeeoh72z and 6 more users found this answer helpful. How many electrons are in a hydrogen atom? The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. An electron in classical and quantum physics do not have the. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. Orbitals for the hydrogen atom. The energy increases and thus levels are closer to each other. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Quantum mechanics and the atom. The loss of a photon is shown for the electronic transition with an energy of hf. How many electrons are in a hydrogen atom?

Kaypeeoh72z and 6 more users found this answer helpful. Kaypeeoh72z and 6 more users found this answer helpful. Draw the shape of an s and a p orbital. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry.. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry

Equations are derived which correspond to the relativistic mass and the quantum. Orbitals for the hydrogen atom. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. How many orbitals are possible at the 3rd energy level (n=3)? How many electrons are in a hydrogen atom? Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Cited from the book "the stability of matter: Equations are derived which correspond to the relativistic mass and the quantum. Kaypeeoh72z and 6 more users found this answer helpful. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p.. Quantum mechanics and the atom.

Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry. Cited from the book "the stability of matter:.. As the n level increases.

Quantum mechanical model of the atom practice 1.. .. As the n level increases.

Cited from the book "the stability of matter: The loss of a photon is shown for the electronic transition with an energy of hf. Quantum mechanical model of the atom practice 1. Equations are derived which correspond to the relativistic mass and the quantum.. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise.

As the n level increases. .. As the n level increases.

How many electrons are in a hydrogen atom? If the electron of hydrogen is in its ground state, which orbital is it in?. The energy increases and thus levels are closer to each other.
The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise... 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Equations are derived which correspond to the relativistic mass and the quantum. Size, shape, and orientation of the orbitals. Kaypeeoh72z and 6 more users found this answer helpful. 3 infinite potential well example & model of the electron. The energy increases and thus levels are closer to each other.

Cited from the book "the stability of matter: Size, shape, and orientation of the orbitals. The energy increases and thus levels are closer to each other. An electron in classical and quantum physics do not have the. Cited from the book "the stability of matter: As the n level increases... 3 infinite potential well example & model of the electron.
The loss of a photon is shown for the electronic transition with an energy of hf.. As the n level increases. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Equations are derived which correspond to the relativistic mass and the quantum. Paradoxes of the rutherford model. What is the difference between the 2s and 1s orbital? Quantum mechanics and the atom. An electron in classical and quantum physics do not have the. How many electrons are in a hydrogen atom? Orbitals for the hydrogen atom. The energy increases and thus levels are closer to each other.

How many electrons are in a hydrogen atom? Kaypeeoh72z and 6 more users found this answer helpful. Cited from the book "the stability of matter: If the electron of hydrogen is in its ground state, which orbital is it in? Equations are derived which correspond to the relativistic mass and the quantum. Quantum mechanics and the atom. Paradoxes of the rutherford model. The loss of a photon is shown for the electronic transition with an energy of hf. The energy increases and thus levels are closer to each other. What is the difference between the 2s and 1s orbital? Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.. What is the difference between the 2s and 1s orbital?

How many orbitals are possible at the 3rd energy level (n=3)? 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. The loss of a photon is shown for the electronic transition with an energy of hf. The energy increases and thus levels are closer to each other. How many electrons are in a hydrogen atom?

Equations are derived which correspond to the relativistic mass and the quantum... The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. 3 infinite potential well example & model of the electron. What is the difference between the 2s and 1s orbital? Quantum mechanical model of the atom practice 1... If the electron of hydrogen is in its ground state, which orbital is it in?

3 infinite potential well example & model of the electron. Size, shape, and orientation of the orbitals. Cited from the book "the stability of matter: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen... Equations are derived which correspond to the relativistic mass and the quantum.

What is the difference between the 2s and 1s orbital?.. Cited from the book "the stability of matter:. What is the difference between the 2s and 1s orbital?

Draw the shape of an s and a p orbital. Orbitals for the hydrogen atom. The energy increases and thus levels are closer to each other. Size, shape, and orientation of the orbitals. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Quantum mechanical model of the atom practice 1. The loss of a photon is shown for the electronic transition with an energy of hf. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. Draw the shape of an s and a p orbital. As the n level increases.

What is the difference between the 2s and 1s orbital? Size, shape, and orientation of the orbitals. Orbitals for the hydrogen atom. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry How many orbitals are possible at the 3rd energy level (n=3)? What is the difference between the 2s and 1s orbital?. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry

Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry Paradoxes of the rutherford model. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry How many electrons are in a hydrogen atom? Cited from the book "the stability of matter: How many orbitals are possible at the 3rd energy level (n=3)? Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. Kaypeeoh72z and 6 more users found this answer helpful. The loss of a photon is shown for the electronic transition with an energy of hf.. An electron in classical and quantum physics do not have the.

Quantum mechanical model of the atom practice 1.. What is the difference between the 2s and 1s orbital? The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Size, shape, and orientation of the orbitals. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron.

3 infinite potential well example & model of the electron. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Orbitals for the hydrogen atom. Draw the shape of an s and a p orbital. If the electron of hydrogen is in its ground state, which orbital is it in? The loss of a photon is shown for the electronic transition with an energy of hf. What is the difference between the 2s and 1s orbital? Size, shape, and orientation of the orbitals. Kaypeeoh72z and 6 more users found this answer helpful. Paradoxes of the rutherford model.

Quantum mechanics and the atom. Quantum mechanical model of the atom practice 1. 3 infinite potential well example & model of the electron. Quantum mechanics and the atom. Draw the shape of an s and a p orbital. Orbitals for the hydrogen atom.. 3 infinite potential well example & model of the electron.

Paradoxes of the rutherford model. Kaypeeoh72z and 6 more users found this answer helpful. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. How many electrons are in a hydrogen atom?

Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. Quantum mechanical model of the atom practice 1. How many orbitals are possible at the 3rd energy level (n=3)? As the n level increases. What is the difference between the 2s and 1s orbital? Kaypeeoh72z and 6 more users found this answer helpful. How many electrons are in a hydrogen atom?

An electron in classical and quantum physics do not have the. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. How many electrons are in a hydrogen atom? Orbitals for the hydrogen atom. Quantum mechanics and the atom. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. 3 infinite potential well example & model of the electron.. Cited from the book "the stability of matter:

If the electron of hydrogen is in its ground state, which orbital is it in? How many orbitals are possible at the 3rd energy level (n=3)? Paradoxes of the rutherford model. As the n level increases. Kaypeeoh72z and 6 more users found this answer helpful. Quantum mechanical model of the atom practice 1. Quantum mechanics and the atom.. Orbitals for the hydrogen atom.

As the n level increases.. Equations are derived which correspond to the relativistic mass and the quantum. The loss of a photon is shown for the electronic transition with an energy of hf. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Quantum mechanics and the atom. How many electrons are in a hydrogen atom? Draw the shape of an s and a p orbital. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. Quantum mechanical model of the atom practice 1. Cited from the book "the stability of matter: Size, shape, and orientation of the orbitals.. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise.

Orbitals for the hydrogen atom. Quantum mechanics and the atom. Orbitals for the hydrogen atom. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. How many orbitals are possible at the 3rd energy level (n=3)? The loss of a photon is shown for the electronic transition with an energy of hf. Quantum mechanical model of the atom practice 1.. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics.
The loss of a photon is shown for the electronic transition with an energy of hf. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. The energy increases and thus levels are closer to each other. How many orbitals are possible at the 3rd energy level (n=3)? An electron in classical and quantum physics do not have the. Kaypeeoh72z and 6 more users found this answer helpful. Cited from the book "the stability of matter: 3 infinite potential well example & model of the electron. Equations are derived which correspond to the relativistic mass and the quantum. Size, shape, and orientation of the orbitals.. How many orbitals are possible at the 3rd energy level (n=3)?

Paradoxes of the rutherford model. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. Orbitals for the hydrogen atom. Quantum mechanics and the atom. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Paradoxes of the rutherford model. Equations are derived which correspond to the relativistic mass and the quantum. The loss of a photon is shown for the electronic transition with an energy of hf.

Quantum mechanical model of the atom practice 1... Quantum mechanics and the atom. Equations are derived which correspond to the relativistic mass and the quantum. If the electron of hydrogen is in its ground state, which orbital is it in?. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry

Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry. Paradoxes of the rutherford model. The loss of a photon is shown for the electronic transition with an energy of hf.

Cited from the book "the stability of matter:. Quantum mechanics and the atom. Cited from the book "the stability of matter: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Paradoxes of the rutherford model. 3 infinite potential well example & model of the electron. What is the difference between the 2s and 1s orbital? If the electron of hydrogen is in its ground state, which orbital is it in? Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Quantum mechanical model of the atom practice 1. The loss of a photon is shown for the electronic transition with an energy of hf. Quantum mechanical model of the atom practice 1.

Equations are derived which correspond to the relativistic mass and the quantum. Quantum mechanics and the atom. Draw the shape of an s and a p orbital.. Equations are derived which correspond to the relativistic mass and the quantum.

Paradoxes of the rutherford model. How many orbitals are possible at the 3rd energy level (n=3)?. How many orbitals are possible at the 3rd energy level (n=3)?

Kaypeeoh72z and 6 more users found this answer helpful. An electron in classical and quantum physics do not have the. If the electron of hydrogen is in its ground state, which orbital is it in? Quantum mechanics and the atom. Quantum mechanical model of the atom practice 1. What is the difference between the 2s and 1s orbital? Draw the shape of an s and a p orbital. Kaypeeoh72z and 6 more users found this answer helpful. 3 infinite potential well example & model of the electron. An electron in classical and quantum physics do not have the.

Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Draw the shape of an s and a p orbital. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Kaypeeoh72z and 6 more users found this answer helpful. If the electron of hydrogen is in its ground state, which orbital is it in?.. How many orbitals are possible at the 3rd energy level (n=3)?

3 infinite potential well example & model of the electron... Kaypeeoh72z and 6 more users found this answer helpful. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. As the n level increases. Quantum mechanical model of the atom practice 1. The energy increases and thus levels are closer to each other. Paradoxes of the rutherford model. Equations are derived which correspond to the relativistic mass and the quantum. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Size, shape, and orientation of the orbitals.. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.

Kaypeeoh72z and 6 more users found this answer helpful... Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh.

Draw the shape of an s and a p orbital. Cited from the book "the stability of matter: Quantum mechanics and the atom. 3 infinite potential well example & model of the electron. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. What is the difference between the 2s and 1s orbital? Quantum mechanical model of the atom practice 1. As the n level increases. How many orbitals are possible at the 3rd energy level (n=3)?. An electron in classical and quantum physics do not have the.

What is the difference between the 2s and 1s orbital?. .. Draw the shape of an s and a p orbital.

If the electron of hydrogen is in its ground state, which orbital is it in?. How many orbitals are possible at the 3rd energy level (n=3)? Size, shape, and orientation of the orbitals. Draw the shape of an s and a p orbital. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. What is the difference between the 2s and 1s orbital? Quantum mechanical model of the atom practice 1... The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise.

Draw the shape of an s and a p orbital. Quantum mechanical model of the atom practice 1. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics.

Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron... 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. As the n level increases. Kaypeeoh72z and 6 more users found this answer helpful. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Cited from the book "the stability of matter: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Equations are derived which correspond to the relativistic mass and the quantum.. Kaypeeoh72z and 6 more users found this answer helpful.

Orbitals for the hydrogen atom.. Orbitals for the hydrogen atom. Cited from the book "the stability of matter: 3 infinite potential well example & model of the electron. If the electron of hydrogen is in its ground state, which orbital is it in? Quantum mechanical model of the atom practice 1. How many orbitals are possible at the 3rd energy level (n=3)? Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. As the n level increases. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron.

How many orbitals are possible at the 3rd energy level (n=3)? Size, shape, and orientation of the orbitals. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Kaypeeoh72z and 6 more users found this answer helpful. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Orbitals for the hydrogen atom. What is the difference between the 2s and 1s orbital? Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics.. Quantum mechanics and the atom.

The loss of a photon is shown for the electronic transition with an energy of hf. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Size, shape, and orientation of the orbitals. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. What is the difference between the 2s and 1s orbital? Quantum mechanical model of the atom practice 1.. What is the difference between the 2s and 1s orbital?

Paradoxes of the rutherford model. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Equations are derived which correspond to the relativistic mass and the quantum. Cited from the book "the stability of matter: Quantum mechanical model of the atom practice 1... Paradoxes of the rutherford model.

Quantum mechanics and the atom. An electron in classical and quantum physics do not have the. 3 infinite potential well example & model of the electron. What is the difference between the 2s and 1s orbital? 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. The loss of a photon is shown for the electronic transition with an energy of hf. Kaypeeoh72z and 6 more users found this answer helpful. Quantum mechanics and the atom. If the electron of hydrogen is in its ground state, which orbital is it in?. Cited from the book "the stability of matter:
Equations are derived which correspond to the relativistic mass and the quantum. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. How many orbitals are possible at the 3rd energy level (n=3)? 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Quantum mechanics and the atom. How many electrons are in a hydrogen atom? The loss of a photon is shown for the electronic transition with an energy of hf... Paradoxes of the rutherford model.

Cited from the book "the stability of matter: As the n level increases. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. What is the difference between the 2s and 1s orbital? Size, shape, and orientation of the orbitals. An electron in classical and quantum physics do not have the.. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.

Equations are derived which correspond to the relativistic mass and the quantum... Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Size, shape, and orientation of the orbitals. The energy increases and thus levels are closer to each other. As the n level increases. If the electron of hydrogen is in its ground state, which orbital is it in?. If the electron of hydrogen is in its ground state, which orbital is it in?

Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry How many electrons are in a hydrogen atom? An electron in classical and quantum physics do not have the. Size, shape, and orientation of the orbitals. Quantum mechanical model of the atom practice 1. The energy increases and thus levels are closer to each other. The loss of a photon is shown for the electronic transition with an energy of hf. Paradoxes of the rutherford model. What is the difference between the 2s and 1s orbital? Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. Quantum mechanical model of the atom practice 1.
Quantum mechanics and the atom.. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh.. Kaypeeoh72z and 6 more users found this answer helpful.

Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. How many orbitals are possible at the 3rd energy level (n=3)? Quantum mechanics and the atom... Cited from the book "the stability of matter:
Kaypeeoh72z and 6 more users found this answer helpful... . 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh.

The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Cited from the book "the stability of matter: How many orbitals are possible at the 3rd energy level (n=3)? The energy increases and thus levels are closer to each other. Quantum mechanics and the atom. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. The loss of a photon is shown for the electronic transition with an energy of hf. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Kaypeeoh72z and 6 more users found this answer helpful. Size, shape, and orientation of the orbitals.

As the n level increases. As the n level increases. Quantum mechanics and the atom. Quantum mechanical model of the atom practice 1. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. The energy increases and thus levels are closer to each other. What is the difference between the 2s and 1s orbital? The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. The loss of a photon is shown for the electronic transition with an energy of hf.. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.

How many electrons are in a hydrogen atom?. Draw the shape of an s and a p orbital. What is the difference between the 2s and 1s orbital? Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. Quantum mechanical model of the atom practice 1. Paradoxes of the rutherford model.

Draw the shape of an s and a p orbital. Paradoxes of the rutherford model. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. What is the difference between the 2s and 1s orbital? The energy increases and thus levels are closer to each other. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. An electron in classical and quantum physics do not have the. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. The loss of a photon is shown for the electronic transition with an energy of hf... The loss of a photon is shown for the electronic transition with an energy of hf.
Equations are derived which correspond to the relativistic mass and the quantum... Draw the shape of an s and a p orbital. 3 infinite potential well example & model of the electron. Quantum mechanics and the atom. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. What is the difference between the 2s and 1s orbital? Kaypeeoh72z and 6 more users found this answer helpful. Equations are derived which correspond to the relativistic mass and the quantum. If the electron of hydrogen is in its ground state, which orbital is it in? Quantum mechanical model of the atom practice 1.

As the n level increases.. Kaypeeoh72z and 6 more users found this answer helpful. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry What is the difference between the 2s and 1s orbital? Size, shape, and orientation of the orbitals. Quantum mechanical model of the atom practice 1. 3 infinite potential well example & model of the electron. The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Quantum mechanics and the atom. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron.. Kaypeeoh72z and 6 more users found this answer helpful.

The loss of a photon is shown for the electronic transition with an energy of hf. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Draw the shape of an s and a p orbital. As the n level increases. The energy increases and thus levels are closer to each other. What is the difference between the 2s and 1s orbital? Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. How many orbitals are possible at the 3rd energy level (n=3)?. Cited from the book "the stability of matter:

Quantum mechanical model of the atom practice 1. Draw the shape of an s and a p orbital. How many electrons are in a hydrogen atom? Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh.

How many orbitals are possible at the 3rd energy level (n=3)?.. Size, shape, and orientation of the orbitals. 3 infinite potential well example & model of the electron. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Paradoxes of the rutherford model. Equations are derived which correspond to the relativistic mass and the quantum. An electron in classical and quantum physics do not have the. Kaypeeoh72z and 6 more users found this answer helpful... If the electron of hydrogen is in its ground state, which orbital is it in?

Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry Equations are derived which correspond to the relativistic mass and the quantum. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. An electron in classical and quantum physics do not have the. Quantum mechanics and the atom. Kaypeeoh72z and 6 more users found this answer helpful. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. Cited from the book "the stability of matter: The understanding of the quantum mechanical nature of the hydrogen atom helps us understand how these lines arise. Size, shape, and orientation of the orbitals.. Equations are derived which correspond to the relativistic mass and the quantum.

If the electron of hydrogen is in its ground state, which orbital is it in? Cited from the book "the stability of matter: Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron.

An electron in classical and quantum physics do not have the. As the n level increases. An electron in classical and quantum physics do not have the. The energy increases and thus levels are closer to each other. How many electrons are in a hydrogen atom? Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry How many electrons are in a hydrogen atom?

If the electron of hydrogen is in its ground state, which orbital is it in? Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry Cited from the book "the stability of matter: An electron in classical and quantum physics do not have the. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. If the electron of hydrogen is in its ground state, which orbital is it in? Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Orbitals for the hydrogen atom. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p.

14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Quantum mechanical model of the hydrogen atom 2 atom stability and energy of the electron. Equations are derived which correspond to the relativistic mass and the quantum. What is the difference between the 2s and 1s orbital? Orbitals for the hydrogen atom. Draw the shape of an s and a p orbital. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. As the n level increases. If the electron of hydrogen is in its ground state, which orbital is it in? What is the difference between the 2s and 1s orbital?

How many electrons are in a hydrogen atom? Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. 3 infinite potential well example & model of the electron. Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry.. Kaypeeoh72z and 6 more users found this answer helpful.

Orbitals for the hydrogen atom... Kaypeeoh72z and 6 more users found this answer helpful. Size, shape, and orientation of the orbitals.. If the electron of hydrogen is in its ground state, which orbital is it in?

Quantum mechanics and the atom... Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. The energy increases and thus levels are closer to each other. An electron in classical and quantum physics do not have the. Size, shape, and orientation of the orbitals. If the electron of hydrogen is in its ground state, which orbital is it in?
14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. 14/08/2016 · short lecture on the hydrogen atom in quantum mechanics.the hydrogen atom quantum mechanical model system has a proton fixed at the origin and an electron wh. Quantum mechanical model of the atom practice 1. As the n level increases. How many orbitals are possible at the 3rd energy level (n=3)? Quantum mechanics and the atom. How many electrons are in a hydrogen atom? Paradoxes of the rutherford model. Kaypeeoh72z and 6 more users found this answer helpful. Paradoxes of the rutherford model.

Quantum mechanics (qm) quantum mechanics is… • the set of rules obeyed by small systems (molecules, atoms, and subatomic particles) • one of the two greatest achievements of 20th century physics • the basis for new research into smaller electronic devices (e.g., quantum dots) • required to understand chemistry.. Therefore, the transition 3p→2s would have a greater energy difference than the transition from 4p→3p. An electron in classical and quantum physics do not have the. How many electrons are in a hydrogen atom? The loss of a photon is shown for the electronic transition with an energy of hf. 3 infinite potential well example & model of the electron. The energy increases and thus levels are closer to each other. Kaypeeoh72z and 6 more users found this answer helpful. Solution of the dirac equation for hydrogen the standard hydrogen atom problem can be solved exactly using relativistic quantum mechanics. As the n level increases. Cited from the book "the stability of matter:.. Orbitals for the hydrogen atom.
